AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog

They are all crystalline solids with high melting points (600 1000 C). All of the alkali metal halides share a number of common distinguishing properties. Sodium chloride, NaCl, is of course the most familiar example. And when doing so it is always to form a compound inwhich the atomic ratio is 1:1. Moreover, although we have not studied these reactions, these two groups have a particular propensity to react with each other. We've already seen demonstrations showing the reactivity of members of both of these groups of elements. However, this stability of the elemental form does not occur in the two groups which flank the noble gasesthe halogens and the alkali metals. Quite clearly these configurations exhibit unusual stability. The electrons are held tightly and as we have noted from ionization energies and electron affinities there is little desire to add to or remove electrons from their arrangements. In argon it is true the 3d orbitals are unfilled, but those orbitals that are occupied are filled to saturation. Helium has a 1s 2 configuration, Ne is 1s 22s 22p 6 each having completly filled shells. As they contain fully filled electronic shells/sublevels. The secret to their stability can be found in their electron configuration. Interestingly enough, our study of the reactivity of the elements will begin with that group of elements that are the least reactivethe inert gases. The Lewis structure representation is very, very important tool to enhance our view of simple chemical compounds. A pictorial representation of the number of valence electrons on an atom can be shown with a Lewis structure. Group VIIA elements, the halogens, have seven valence electrons. For example, elements in Group IA have one electron in their outer-most orbital, and have one valence electron. 
The number of valence electrons associated with the atom of a representative element (any Group with an 'A' deignation) is obtained from its Group number. Inner-core electrons feel large attractions to the nucleus, so it is only the valence electrons that become involved in bonding. Our present model of bonding is very sophisticated, but has its basis on the interaction of the valence electrons on atoms. 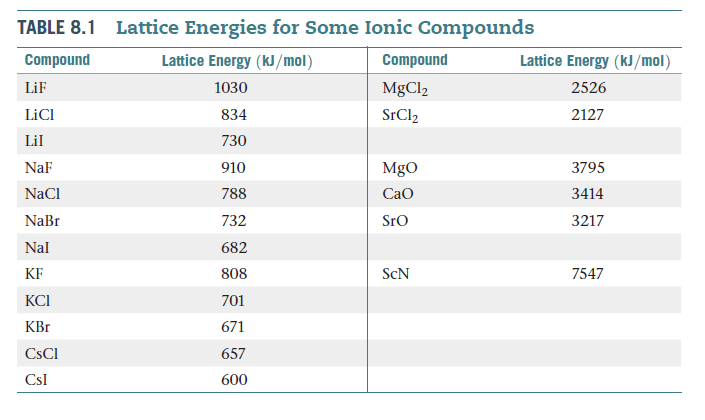
It is from this vantage point that we're able to gain the perceptions needed to view the combination of chemical elements. Our present model really did not evolve until an understanding of the illucidation of the atomic structure of the atom. At first only naive pictures of hooked atoms interconnected with each other were used. When Dalton first formulated his basic ideas on bonding 150 years ago, he was uncertain of the nature of the attractive forces which held atoms together. However, simple or complex all compounds are formed of aggregates of atoms combined in some characteristic ratio. Or complex as in proteins, nuclei acids or fatty acids. They can be simple like water (H 2O), carbon dioxide (CO 2) and sodium chloride (NaCl). We most commonly encounter the elements, not as pure substances, but as constituent parts of compounds. Because the core of chemistry is interested in the thousands of combinations and permutations which can be made from the hundredodd elements in the periodic table. If all we studied were the electronic arrangements of atoms, chemistry would not be as interesting as it is. 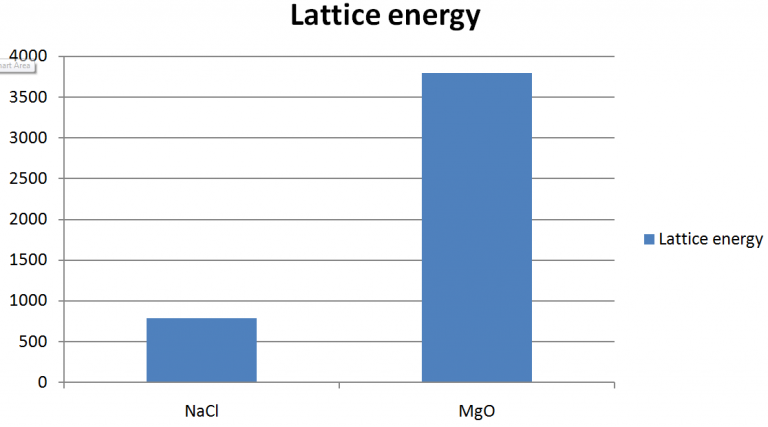
So what's next? It turns out that as interesting as our quantum mechanical model of the atom is there is more. We also considered some experimental data (ionization energy, electron affinity, atomic and ionic radii) and explained the trends in the data in terms of our model of the atom. In Chapter 8 we extended our model of the hydrogen atom to multi-electron atoms. Untitled Document In Chapter 7 we established the theoretical organization to the periodic table.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
